|
See the explanation in the paragraph above (b) and (c) show two other ways to represent a water molecule, other than the molecular formula, H 2O, and the structural formula, H-O-H. Similarly, carbon dioxide, which contains one carbon atom and two oxygen atoms in each molecule, is written as CO2. molecular versus formula weights as it is to understand the concept of the mole, because one can speak of 1 mole of carbon, carbon dioxide, and sodium chloride. Each hydrogen atom (group 1) has 1 valence electron, carbon (group 14) has 4 valence electrons, and oxygen (group 16) has 6 valence electrons, for a total of (2) (1) + 4 + 6 12 valence electrons. To tell if CO2 (Carbon dioxide) is ionic or covalent (also called molecular) we look at the Periodic Table that and see that C is a. For example, bromine, Br 2 \text CH 4 start text, C, H, end text, start subscript, 4, end subscript -oxygen, and nitrogen-which only have London dispersion forces of attraction between the molecules-freeze at very low temperatures.\) Water: a polar covalent molecule (a) shows two polar covalent bonds in the water molecule. Determine the total number of valence electrons in the molecule or ion. The more electrons a molecule has, the stronger the London dispersion forces are. For each of the following questions, determine whether the compound is ionic or covalent and write the appropriate formula for it. Classify C 60, BaBr 2, GaAs, and AgZn as ionic, covalent, molecular, or metallic solids and then arrange them in order of increasing melting points. 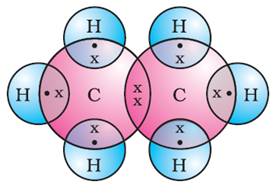
Identify the bonding in Br 2 as ionic or.  
London dispersion forces, under the category of van der Waal forces: These are the weakest of the intermolecular forces and exist between all types of molecules, whether ionic or covalent-polar or nonpolar. The actual melting points are C 6 (CH 3) 6, 166☌ Zn, 419☌ RbI, 642☌ and Ge, 938☌. carbon and oxygen atoms are connected by covalent bonds (shown above).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
